
Ice VIII substructure
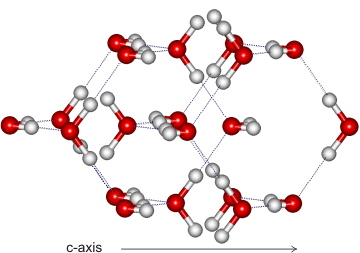
Ice-eight (ice VIII) forms from ice-seven (ice VII) on lowering its temperature (see Phase Diagram). The hydrogen bonding is ordered and fixed as ice-seven undergoes a proton disorder-order transition to ice eight when cooled at about 5 °C; ice-seven and ice-eight having identical structures apart from the proton ordering. The proton ordering causes a slight distortion in the ice-seven cubic lattice (a,b slightly shorter, c slightly longer) resulting in a tetragonal crystal structure (I41/amd, 141; Laue class symmetry 4/mmm) where all of the water molecules are hydrogen-bonded to four others, two as a donor and two as an acceptor. Similar to ice-seven, ice-eight consists of two interpenetrating cubic ice lattices in which each oxygen atom is surrounded by its eight nearest neighbors but is connected to only four tetrahedrally coordinated neighbors with hydrogen bonds.
Ice VIII substructure variant
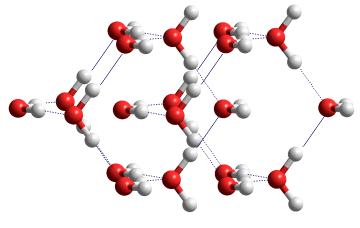
The molecular dipoles in these sublattices point in opposite directions along the c axis, making the crystal antiferro-electric. The bulk modulus and equation of state of ice VIII have been described [3300].
A ferroelectric variant of this structure may exist (see left). In this crystal, the molecular dipoles in the sub-lattices point in the same direction along the c axis, making the crystal ferroelectric (space group P42nm) [2358]. This structure is slightly less stable (≈ 1 kJ ˣ mol−1 ) than the antiferroelectric ice VIII but is likely to exist in domains at high pressure, particularly in an applied electric field [2358].
Ice eight

Ice VIII has a density of about 1.66 g cm−3 (at 8.2 GPa and 223 K [8]), which is less than twice the cubic ice density as the intra-network O····O distances are longer to allow for the interpenetration. Ice-eight's molar volume is slightly smaller (by 0.65 mm3 mol−1) than ice-seven along the phase transition line. Ice-eight has triple points with ice-six and ice-seven (5 °C, 2.1 GPa) and ice-seven and ice-ten (100 K, 62 GPa). The relative permittivity (dielectric constant) of ice-eight is about 4. An equation of state for ice VII and its neighbors has been described up to 80 GPa at low temperatures [4435].
The crystal (shown on the right) has cell dimensions a, b = 4.4493 Å, c = 6.413 Å (90º, 90º, 90º; D2O, at 2.6 GPa and 22 °C [362]), containing eight water molecules per unit cell. All molecules experience identical molecular environments. As the H-O-H angle does not vary much from the isolated molecule, the hydrogen bonds are not straight (although shown so in the figures).
Interactive Jmol structures are given.
Home | Site Index | Phase Diagram | Ices, introduction | Ice-Ih | Ice-Ic | Ice-Isd | II | III | IV | V | VI | VII | IX | X | XI | XII | XIII | XIV | XV | XVI| XVII | XVIII | Amorphous ice | LSBU | Top
This page was established in 2002 and last updated by Martin Chaplin on 23 February, 2022