
Gracilaria brevis

Agar dissolves in hot water to form gels on cooling.
Agar (E406) is a scaffolding polysaccharide prepared from the same family of red seaweeds (Rhodophycae) as the carrageenans. It is commercially obtained from species of Gelidium and Gracilariae
-(1-3)-beta-D-galactopyranose-(1-4)-3,6-anhydro-alpha-L-galactopyranose unit
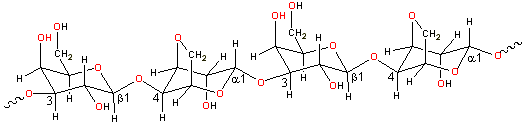
Agar consists of a mixture of agarose and agaropectin. Agarose
is a linear polymer, of relative molecular mass (molecular weight) about 120,000, based on
the -(1![]() 3)-β-D-galactopyranose-(1
3)-β-D-galactopyranose-(1![]() 4)-3,6-anhydro-α-L-galactopyranose
unit; the major differences from carrageenans being the presence of L-3,6-anhydro-α-galactopyranose
rather than D-3,6-anhydro-α-galactopyranose
units and the lack of sulfate groups. Agaropectin is a heterogeneous
mixture of smaller molecules that occur in lesser amounts. Their
structures are similar but slightly branched and sulfated, and they
may have methyl and pyruvic acid ketal substituents. They gel poorly
and may be removed easily from the excellent gelling agarose molecules
by using their charge. Alkaline
treatment that converts any L-galactose-6-sulfate to 3,6-anhydro-L-galactose improves the quality of the agar. [Back to Top
4)-3,6-anhydro-α-L-galactopyranose
unit; the major differences from carrageenans being the presence of L-3,6-anhydro-α-galactopyranose
rather than D-3,6-anhydro-α-galactopyranose
units and the lack of sulfate groups. Agaropectin is a heterogeneous
mixture of smaller molecules that occur in lesser amounts. Their
structures are similar but slightly branched and sulfated, and they
may have methyl and pyruvic acid ketal substituents. They gel poorly
and may be removed easily from the excellent gelling agarose molecules
by using their charge. Alkaline
treatment that converts any L-galactose-6-sulfate to 3,6-anhydro-L-galactose improves the quality of the agar. [Back to Top ![]() ]
]
The gel
network of agarose contains double helices formed from left-handed
threefold helices. The presence
of water molecules bound to the inside of the double-helical cavity stabilizes these double helices [508].
Exterior hydroxyl groups allow aggregation of up to 10,000 of these
helices to form suprafibers. Spectrophotometric investigation of agar gel formation has suggested that the gel formation occurs over a much narrower temperature range than its collapse [4048]. [Back to Top ![]() ]
]
Agar is insoluble in cold water but dissolves in boiling water to give random coils. Gelation is reported to follow a phase separation process [501a] (although these findings are disputed [501b]) and association on cooling (≈ 35 °C), forming gels with up to 99.5% water and remaining solid up to about 85 °C. Agar has significant use in microbiological media. It is not easy for microorganisms to metabolize and forms clear, stable, and firm gels, but it finds use in icings, glazes, processed cheese, jelly sweets, and marshmallows. It may be used in tropical countries and by vegetarians as a substitute for gelatin. Agar gel can be used to selectively clean the surface of sensitive artworks and historical artifacts by the gradual removal of any soiling layer through the slow release of water (syneresis), diffusion of water into the porous substrate, and the withdrawal of the dirty water (evaporation, capillarity, and osmosis) [3343].
Interactive structures are available (Jmol). [Back to Top ![]() ]
]
Home | Site Index | Hydrocolloids | Polysaccharide hydration | hydrogen-bonding | LSBU | Top
This page was established in 2003 and last updated by Martin Chaplin on 8 November, 2021